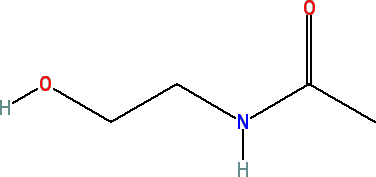
Acetamide MEA
Safety Information
The Food and Drug Administration (FDA) includes acetamide MEA (as N-acetyl ethanolamine) on its list of approved indirect food additives. It is permitted for use as a component of adhesives having incidental contact with food. The safety of acetamide MEA has been assessed by the Expert Panel for Cosmetic Ingredient Safety. The Expert Panel evaluated the scientific data and concluded that acetamide MEA is safe in rinse-off products, and in leave-on products at concentrations not to exceed 7.5%. Cosmetics and personal care products containing acetamide MEA should not contain nitrosating agents or significant amounts of free acetamide.
Acetamide MEA exhibited no acute toxicity following dermal exposure. In ocular irritation studies, 70% acetamide MEA and cosmetic formulations containing 1.3% acetamide MEA were not irritating to the eyes. Only mild skin irritation occurred following a 24 hour skin exposure to undiluted acetamide MEA. In a sensitization test, acetamide MEA was classified as a nonsensitizer when tested at a concentration of 5.0%.
Neither primary irritation nor sensitization reactions to 7.5% acetamide MEA were observed in a human repeated insult patch test. Acetamide MEA was not nonmutagenic in bacterial tests. The Panel noted that in the presence of nitrosating agents, acetamide MEA may form N-nitroso compounds, which are potential carcinogens. Acetamide may be a minor impurity in acetamide MEA. Therefore, the Expert Panel concluded that cosmetics and personal care products containing acetamide MEA should not containe nitrosating agents or significant amounts of free acetamide.
More information about nitrosamines.
Acetamide MEA may be used in cosmetics and personal care products marketed in Europe according to the general provisions of the Cosmetics Regulation of the European Union.